TM 5-3820-245-14&P
of these. Chlorides, sulfates, magnesium and calcium
contribute to cavitation erosion. Cavitation erosion is
are among the materials which make up dissolved
caused by the collapse of bubbles (vapor pockets)
solids. Water, within the limits specified in Table 1 is
formed at the coolant side of an engine component. The
satisfactory as an engine coolant when proper inhibitors
collapse results from a pressure differential in the liquid
are added. The procedure for evaluating water intended
caused by the vibration of the engine part. As bubbles
for use in a coolant solution is shown in Table 2.
collapse, they form pin points of very high pressure.
Over a period of time, the rapid succession of millions of
CORROSION INHIBITORS VITAL
tiny bursting bubbles can wear away (erode) internal
A corrosion inhibitor is a water-soluble chemical
engine surfaces.
compound which protects the metallic surfaces of the
Components such as fresh water pump impellers and
cooling system against corrosive attack. Some of the
cylinder liners are especially susceptible to cavitation
more commonly used corrosion inhibitors are chromates,
erosion. In extreme cases their surfaces can become so
borates, nitrates, nitrites and soluble oil. (Soluble oil is
deeply pitted that they appear to be spongy, and holes
not recommended as a corrosion inhibitor). Depletion of
can develop completely through them.
all types of inhibitors occurs through normal operation.
Therefore, strength levels must be maintained by the
Chromates
addition of inhibitors at prescribed intervals.
Sodium chromate and potassium dichromate are two of
The importance of a properly inhibited coolant cannot be
the best and most commonly used water system
overstressed. A coolant which has insufficient inhibitors,
corrosion inhibitors.
Care should be exercised in
the wrong inhibitors, or-worse-no inhibitors at all invites
handling these materials due to their toxic nature.
the formation of rust and scale deposits within the
Chromate inhibitors should not be used in antifreeze
cooling system. Rust, scale, and mineral deposits can
solutions. Chromium hydroxide, commonly called "green
wear out water pump seals and coat the walls of the
slime", can result from the use of chromate inhibitors
cylinder block water jackets and the outside walls of the
with antifreeze. This material deposits on the cooling
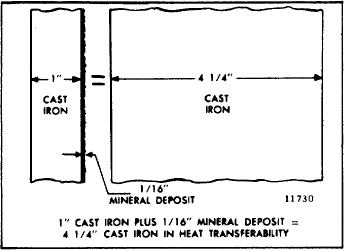
cylinder liners. As these deposits build up, they insulate
system passages and reduces the heat transfer rate
the metal and reduce the rate of heat transfer. For
(Fig. 1) which results in engine overheating. Engines
example, a 1/16" deposit of rust or scale on 1" of cast
which have operated with a chromate-inhibited water
iron is equivalent to 4-1/4" of cast iron in heat
must be chemically cleaned before the addition of
transferability (Fig. 1).
antifreeze. A commercial heavy duty descaler should be
An engine affected in this manner overheats gradually
used in accordance with the manufacturer's
over a period of weeks or months. Liner scuffing,
recommendation for this purpose.
scoring, piston seizure and cylinder head cracking are
Soluble Oil
the inevitable results. An improperly inhibited coolant
Soluble oil has been used as a corrosion inhibitor for
can also become corrosive enough to "eat away" coolant
many years. It has, however, required very close
passages and seal ring grooves and cause coolant
attention relative to the concentration level due to
leaks to develop. If sufficient coolant accumulates on
adverse effects on heat transfer if the concentration
top of a piston, a hydrostatic lock can occur while the
exceeds 1% by volume. For example: 1.25% of soluble
engine is being started. This, in turn, can result in a bent
oil in the cooling system increases fire deck
connecting rod. An improperly inhibited coolant can also
temperatures 6% and a 2.50% concentration raises fire
Soluble oil is not
deck temperature up to 15%.
recommended as a corrosion inhibitor.
Non-Chromates
Non-chromate inhibitors (borates, nitrates, nitrites, etc.)
provide corrosion protection in the cooling system with
the basic advantage that they can be used with either
water or a water-and-antifreeze solution.
INHIBITOR SYSTEMS
An inhibitor system is a combination of chemical
compounds which provide corrosion protection, pH
control and water-softening ability. Corrosion protection
is discussed under the heading Corrosion Inhibitors Vital.
The pH control is used to maintain an acid-free solution.
The water-softening ability deters formation of mineral
Fig. 1 - Heat Transfer Capacity
deposits. Inhibitor systems are available in various
forms such as coolant filter
PAGE 165



